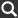pH Levels in Drinking Water
What Is A pH Reading?
The initials pH are used as the measurement of the acidity or alkalinity of a substance. It is sometimes denoted as the “Potential Hydrogen.”
For example, pure water has a pH value of 7 and is considered neutral. As the pH value lowers, the water is considered more acidic and as it elevates above 7 it is considered more alkaline. Standards for drinking water suggest a pH value of between 6.5 and 8.5.
How Does The pH Determine If My Water Is Hard Or Soft?
Water is considered soft as the level drops toward the lower side of 6.5 and out of the recommended range. Soft water can cause the following issues:
- Association with higher levels of metals such as iron, manganese, copper, and lead and the health risks associated with each.
- Acidity can cause premature damage to metal piping.
- Aesthetic problems such as a metallic or sour taste.
- Staining of laundry and staining of sinks and drains, typically blue green.
Water is considered hard when the pH level is above 8.5. Hard water does not pose a health risk, but can cause aesthetic problems such as:
- Bitter taste to the water and beverages such as tea or coffee.
- Scales on piping and fixtures that reduces the internal diameter and can reduce pressure.
- Deposits on dishes and tubs and sinks.
- Lack of lather when using soaps for bathing or detergents in laundry.
- Build up on coils in water heaters can reduce efficiency.
How Do I Treat The pH Levels?
The primary way is through installation of an Acid Neutralizer.
The neutralizer prevents the water from reacting with the plumbing and contributing to electrolytic corrosion. Installation of a water softening system has been effective in raising the pH level in drinking water however it is not actual treatment for pH.
If you are concerned about the pH levels in your drinking water, contact one of our specialists to discuss your water testing and treatment options.
Water Testing Options

Private Well Testing
Are you buying or selling a home with a private well? ATS makes sure you meet your local well testing requirements.
Find out more
Home Water Testing
Our EZ Water Tests provide a complete water testing solution as well as testing for particular contaminants. Our water testing experts are trained to help you with your particular situation.
Find out moreWater Doctor
If you think you have a problem with your water, our water doctor will help you identify the source and if it should be treated.
Find out more